SISTM
Statistics in systems biology and translationnal medicine


Objectives
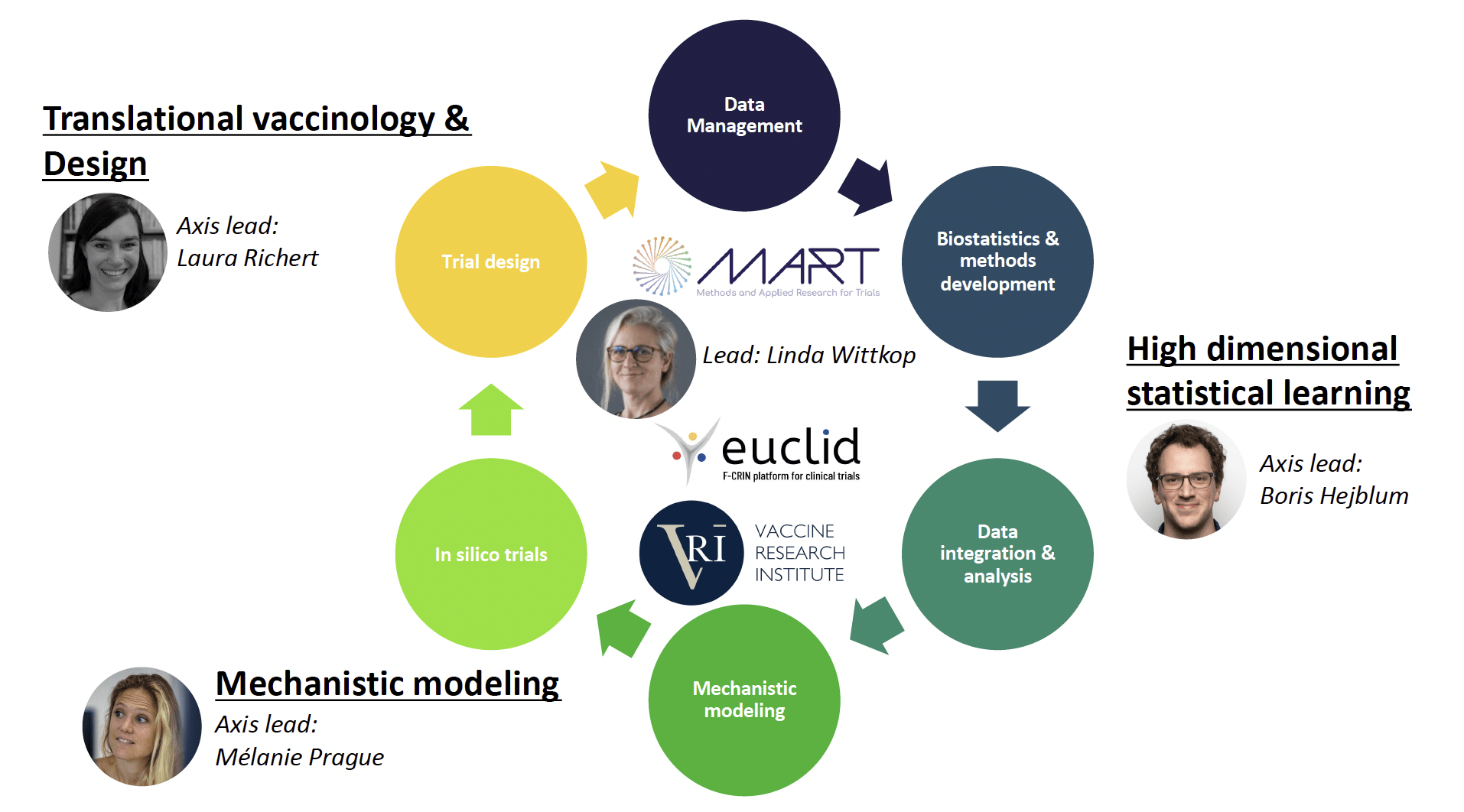
The two main objectives of the SISTM team are :
1) to accelerate the development of vaccines by analysing all the information available in early clinical trials and optimizing new trials
2) to develop new statistical methods to analyse and model large high-throughput data.
Research area
The relevant information is extracted from large omics data, and this signal is then incorporated into mechanistic models, thanks to prior biological knowledge, to estimate their parameters. Those models can then inform the optimal vaccine strategies to be evaluated in the next clinical trials. Such in silico trials, allowing for optimized clinical trial designs and personalized strategies, in turn generate new data that require and feed anew the above methodological developments.
Downloads
For Lasso Estimation of Conditional Logistic
Regression Models for matched case-control and case-crossover studies
2025 Key publications
Ferte T, Ba K, Dan D, Legrand, Jouhet V, Thiebaut R, Hinaut X, Hejblum BP. Reservoir Computing in R: a Tutorial for Using reservoirnet to Predict Complex Time-Series. Computo. 2025. https://openreview.net/forum?id=6kZQTZsHl5
Ferte T, Dutartre D, Hejblum B, Griffier R, Jouhet V, Thiebaut R, Hinaut X, Legrand P. Optimizing Reservoir Computing with Genetic Algorithm for High-Dimensional SARS-CoV-2 Hospitalization Forecasting: Impacts of Genetic Algorithm Hyperparameters on Feature Selection and Reservoir Computing Hyperparameter Tuning. In: Legrand P, Liefooghe A, Lepagnot J, Monmarché N, Idoumghar L, Pallez D, et al., editors. Artificial Evolution. Cham: Lecture Notes in Computer Science; 2026. p. 138-51. https://doi.org/10.1007/978-3-032-07998-5_10
Ganser I, Paireau J, Buckeridge DL, Cauchemez S, Thiebaut R, Prague M. Comparative evaluation of methodologies for estimating the effectiveness of non-pharmaceutical interventions in the context of COVID-19: a simulation study. Am J Epidemiol. 2025. https://doi.org/10.1093/aje/kwaf035
Guerra-Adames A, Avalos-Fernandez M, Doremus O, Gil-Jardine C, Lagarde E, editors. Uncovering Judgment Biases in Emergency Triage: A Public Health Approach Based on Large Language Models. Machine Learning for Health (ML4H); 2025 2025/02/17/: PMLR. https://proceedings.mlr.press/v259/guerra-adames25a.html
Hughes A, Parast L, Thiebaut R, Hejblum BP. RISE: Two-Stage Rank-Based Identification of High-Dimensional Surrogate Markers Applied to Vaccinology. Stat Med. 2025;44(20-22):e70241. https://doi.org/10.1002/sim.70241
Kone C, Kaufmann E, Richert L. Bandit Pareto Set Identification in a Multi-Output Linear Model. Proceedings of Machine Learning Research; Jul 62025. p. 1189-97. https://doi.org/10.48550/arXiv.2507.04255
Luong Nguyen LB, Magloire L, Francois A, Billard D, Priou F, Arrondeau J, Linassier C, Ben Ghezala I, Gross-Goupil M, Charles J, Dohollou N, Vanhems P, Cracowski C, Leroi AM, Laine F, Galtier F, Barthelemy K, Priet S, Gharib M, Chalouni M, Barquin A, Loubet P, de Lamballerie X, Launay O, Wittkop L, Blay J-Y, Spano J-P. Humoral immune response to Covid-19 vaccination in patients with cancer – Results from the ANRS0001S COV-POPART study. Vaccine. 2025;63:127633. https://doi.org/10.1016/j.vaccine.2025.127633
Mitard de Girardier A, Herate C, Marlin R, Donati F, Rahou Y, Bossevot L, Sconosciuti Q, Cavarelli M, Bosquet ND, Relouzat F. Exposure history shapes SARS-CoV-2 viral dynamics in Non-Human Primates and provides insights into correlates of protection against infection and transmission. Philos Trans R Soc B Biol Sci. 2025.In press https://inserm.hal.science/inserm-05291797v1
Thiebaut R, Lhomme E, Hocini H, Pellegrin I, Boizard-Moracchini A, Duvignaud A, Perpere M, Huchon M, Prague M, Lacabaratz C, Surenaud M, Anglaret X, Malvy D, Hejblum B, Group Cs, Levy Y. Self-Collected Finger-Prick Blood for Gene Expression Profiling: Unveiling Early Immune Responses in Mild COVID-19. medRxiv. 2025. https://doi.org/10.1101/2025.05.06.25327063
Xia Y, Alexandre M, Thiébaut R, Maheu-Giroux M, Prague M. Defining Optimal Vaccine Features for Pandemic Preparedness: An Individual-Based Model Bridging Within- and Between-Host Dynamics. Bull Math Biol. 2025. https://papers.ssrn.com/abstract=5205051
Last News


THESIS DEFENCE : THOMAS FERTE
Non classé
Organisation of ‘journées de la statistique’ Annual conference of Société Francaise de Statistique
Non classé
Two BPH researchers obtain their HDR: Boris Hejblum and Mélanie Prague
Awards & nominations
Launch of CAP Digital Health within the framework of the call for expressions of interest "Skills and Future Professions"
Non classé
Covid-19 : Etude de l'efficacité des mesures instaurées contre le virus en France
Press
The BPH SISTM team is accredited by both Inria & Inserm
New project
More than 3 M€ for our project UB 2030 CAP Digital Health labellized by the call AMI CMA "Skills and future professions" of the 2030 France national plan
Awards & nominationsMembers
Avalos FernandezMarta
Associate Professor in BiostatisticsBAKALIDOU
Etudiant en thèseClaironQuentin
Chargé de Recherche INRIAColajanniAntonin
Etudiant en ThèseCrépinLisa
Étudiante en thèseDarmignySandrine
Assistante administrativeDelrieuEmie
Etudiante en thèseEL HAJJZeinab
Ingénieure de rechercheEl KarchiJad
Ingénieur R&DFALLETSARA
Ingénieur d'étudesFerteThomas
Etudiant en thèseGounaDaniela
DoctoranteHejblumBoris
Chargé de Recherche INSERMHivertBenjamin
Post-doctorant / Post-doctoral fellowHosteinsCéline
Étudiante en thèseHUGHESArthur
Etudiant en thèseLAVALQuentin
Ingénieur d'étudesLhommeEdouard
MD, PhD - MCU-PH Santé PubliqueLiZhe
DoctoranteLunelPerrine
Etudiante en thèseNAFTI MAHERZIMyriam
Data ManagerNODATakashi
DoctorantOttaviAnton
Coordinateur de Projets InternationauxPalAnnesh
DoctorantPragueMélanie
Chargé de Recherche InriaRichertLaura
MD, PhD - PU-PH Santé PubliqueRuizAnne-Andrée
Ingénieure d'études BiostatisticienneSAVELHelene
Etudiante en thèseSegalasCorentin
Research Fellow in statisticsThiebautRodolphe
Directeur d'équipeTrầnNam-Anh
PhD studentWittkopLinda
MD PhD - PU-PH Santé Publique